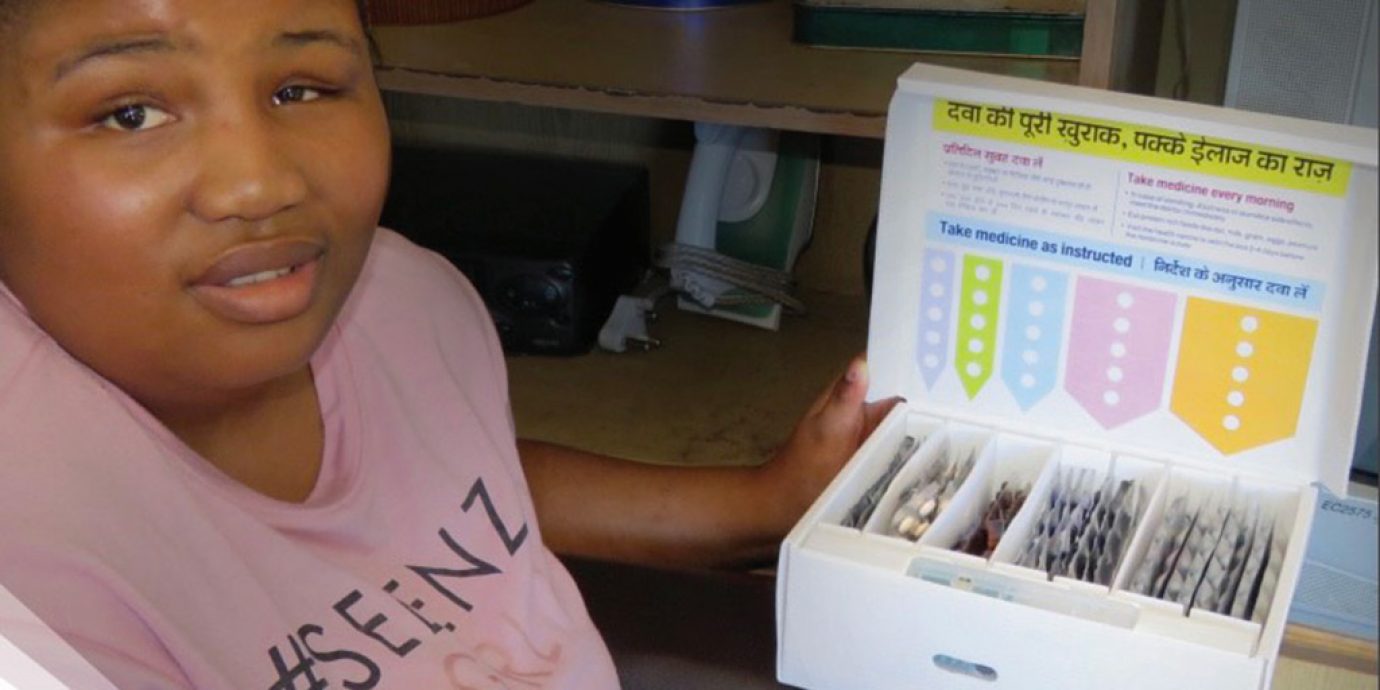
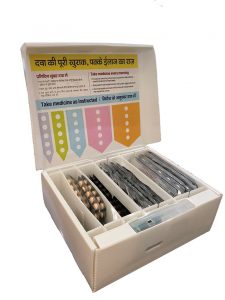
Stop TB Partnership’s Global Drug Facility (GDF) and the Unitaid-funded Adherence Support Coalition to End TB (ASCENT project) announce the inclusion of the first digital adherence technology (DAT) in the GDF’s product catalog: the Wisepill evriMED smart medication container. This marks a turning point in efforts to improve people-centered tuberculosis (TB) treatment.
The introduction of innovative technologies, such as the smart medication container, supports real-time self-administered treatment.
Every time a TB-affected person opens the container to take his or her medicines, the embedded device automatically logs the daily dose. If the container is not opened for multiple days, it may mean some doses were missed. The healthcare provider will be notified and they can then work with the TB-affected person to develop a personalized treatment support plan designed to help him or her complete treatment.
Multiple barriers can prevent affected people from completing their treatment. This can be due to long and complicated treatment regimens that involve the daily intake of medicines over the course of six months to two years. Not completing a treatment course can result in relapse of the disease and increased resistance to TB medicines – meaning more complicated and expensive medicines and regimens may be needed to treat the affected person successfully.

People-centered care
DATs’ advent is part of a shift towards people-centered care, enabling people affected by TB to take charge of their treatment. This is even more relevant now during the global COVID-19 pandemic, as many people face challenges in reaching clinical care and require home-based treatments. A recent survey by global civil society and TB affected communities on the impact of COVID-19 on TB found that up to 50% of survey respondents in Kenya reported difficulties finding transport to care facilities.
The inclusion of the Wisepill evriMED smart medication container in the GDF catalog marks a new chapter in the fight against TB. Until now, access to DATs had been limited, as large-scale procurement mechanisms for the products did not exist.
“For a long time now, the Stop TB Partnership is advocating for the need for innovative solutions to treat and cure people with TB. In these challenging times of the COVID-19 pandemic, I am even more proud to see health technology solutions seeing their way onto the GDF catalog to make sure there is no stopping in our progress against TB,” said Dr. Lucica. Ditiu, Executive Director, Stop TB Partnership.
First procurement
In parallel, the ASCENT project is launching a Total Cost of Ownership tool, which can support countries to plan and budget for the adoption and scale-up of DAT use. The ASCENT project, which is funded by Unitaid and led by KNCV Tuberculosis Foundation, is the first to procure DATs through GDF; 5,000 Wisepill smart medication containers will support persons affected by TB in Ethiopia, the Philippines, South Africa, Tanzania, and Ukraine.
“Digital adherence technologies can provide complementary support to ensure patients take ownership of their care; it equally provides health care workers the opportunity for prioritization of individualized support,” said Dr. Mustapha Gidado, Executive Director, KNCV Tuberculosis Foundation.
“Innovative tools with the potential to simplify access to treatments for people affected by TB are crucial game changers. They can radically improve the way health workers deliver treatments, improve patients’ adherence, and ultimately save lives,” said Dr Philippe Duneton, Executive Director of Unitaid.
Lessons learned on the implementation, acceptability, feasibility, and effectiveness of different DATs in relation to treatment outcomes are forthcoming from the Stop TB Partnership’s TB REACH Wave 6 demonstration projects conducted in multiple countries and involving varied populations. As well as the Treatment Adherence Project, funded by the Bill & Melinda Gates Foundation.
“A critical enabler of the uptake of improved patient-centered digital adherence technologies is an easy and efficient way for country programs to procure the new tools. It’s exciting to see the Global Drug Facility step forward to address this need and to provide this critical help to country programs,” Daniel Chin, Deputy Director, TB-HIV Program Strategy Team, Bill and Melinda Gates Foundation.
For more information, please see the GDF Product Catalog (available here: http://stoptb.org/assets/documents/gdf/drugsupply/GDFProductCatalog.pdf) and the Technical Information Note (available here: http://stoptb.org/assets/documents/gdf/drugsupply/TIN_SMCK.pdf). To place an order, please follow this link: http://stoptb.org/gdf/placeOrder.asp or contact gdf@stoptb.org.
About the Stop TB Partnership

The Stop TB Partnership is a unique United Nations hosted entity based in Geneva, Switzerland, committed to revolutionizing tuberculosis (TB) space to end the disease by 2030. The organization aligns more than 2,000 partners worldwide to promote cross-sectoral collaboration. The Stop TB Partnership’s various teams and initiatives take bold and smart risks to identify, fund and support innovative approaches, ideas, and solutions to ensure the TB community has a voice at the highest political levels and that all TB affected people have access to affordable, quality, and people-centered care. Learn more at www.stoptb.org, and follow us at @StopTB.
About the Global Drug Facility
GDF is the world’s largest procurer of TB medicines and diagnostics. Since its creation in 2001, GDF has facilitated access to TB medicines and diagnostics in more than 140 countries, making quality-assured treatments available to over 32 million people with TB. In 2019 alone, GDF delivered nearly USD264 million worth of TB medicines and diagnostics to 116 countries. GDF has led the introduction of advanced diagnostics and supplies, all-oral regimens for DR-TB as recently recommended by WHO, child-friendly medicines for drug-sensitive TB and shorter regimens for multidrug-resistant TB. Since 2012, GDF has secured price reductions of over 50 percent for most second-line TB medicines, primarily by reducing risk to suppliers and minimizing their transaction costs. Between Jan 2019 – Mar 2020, GDF saved National TB Programs approximately USD53 million: USD33 million in medicine price reductions from competitive tenders; USD15.3 million by readjusting inaccurate quantification/order numbers; and USD4.2 million by allowing flexibility to cancel and postpone previously paid orders.
Over the last decade, GDF’s efforts have spurred a more than five-fold increase in the number of companies producing TB products—from just five in 2007 to 28 in 2019, and an 11-fold rise of quality-assured products from eight in 2007 to 89 in 2019. GDF’s approach to bundle procurement with technical assistance on quantification and supply planning allows GDF to monitor for risks of future stockouts via national early warning systems and take necessary action to avoid stockouts.
About the ASCENT project

The ASCENT project runs from 2019 until the end of 2022. This project is made possible thanks to Unitaid’s funding and support. Unitaid accelerates access to innovative health products and lays the foundations for their scale-up by countries and partners. ASCENT is led by KNCV Tuberculosis Foundation in consortium with the Aurum Institute, London School of Hygiene and Tropical Medicine and PATH. ASCENT focuses on improving adherence and treatment outcomes for TB patients and is implemented in Ethiopia, Ukraine, Tanzania, the Philippines and South Africa. By leveraging today’s smart information and mobile communication technologies, the ASCENT project aims to support TB patients with their treatment in a modern and more effective way through the use of digital adherence technologies such as smart pillboxes, medication sleeves and video supported treatment. By generating evidence, establishing a global market and engaging stakeholders, ASCENT aims to make future scale-up possible so that these digital innovations can be available to all TB patients worldwide.